GS-SMD server
The membranous γ-secretase (GS) complex is performs cleavage of more than 170 substrates, including Amyloid Precursor Protein (APP). The sequential cuts (trimming) of APP leads to formation of amyloidogenic peptides Aβ40 and Aβ42, that can be found in brains of Alzheimer’s disease (AD) patients, in the form of amyloid plaques. The atomistic mechanism of the intramembrane cleavage is not fully known, and especially not known for other than APP substrates of GS. The GS-SMD server allows to perform the steered molecular dynamics (SMD) simulations of many substrates of GS in the binding site of this proteolytic complex. During SMD simulations one can study preferences for making the cleavage at a specific location in the substrate sequence by unfolding the C-terminus of the substrates. In GS-SMD server one can also make mutations in the substrate as well as in the complex to study their influence on the cleavage process. Such studies can be helpful for understanding of this complicated process and also for designing drugs for selective blocking of GS.
The interactive dendrogram of all known γ-secretase substrates based on their transmembrane (TM) sequences. The chart shows sequence similarities among all known substrates. Click on the substrate's UniProt ID to see the information about this substrate. The sequence logo shown below presents the sequences of substrates, each of 39 amino acid length, aligned by the cleavage site, which is No. 0 on the X axis, and hydrophobicity of TM region if the cleavage site is not determined yet. The transmembrane regions can differ among the substrates but approximately they are located between residues No. -19 and 2.
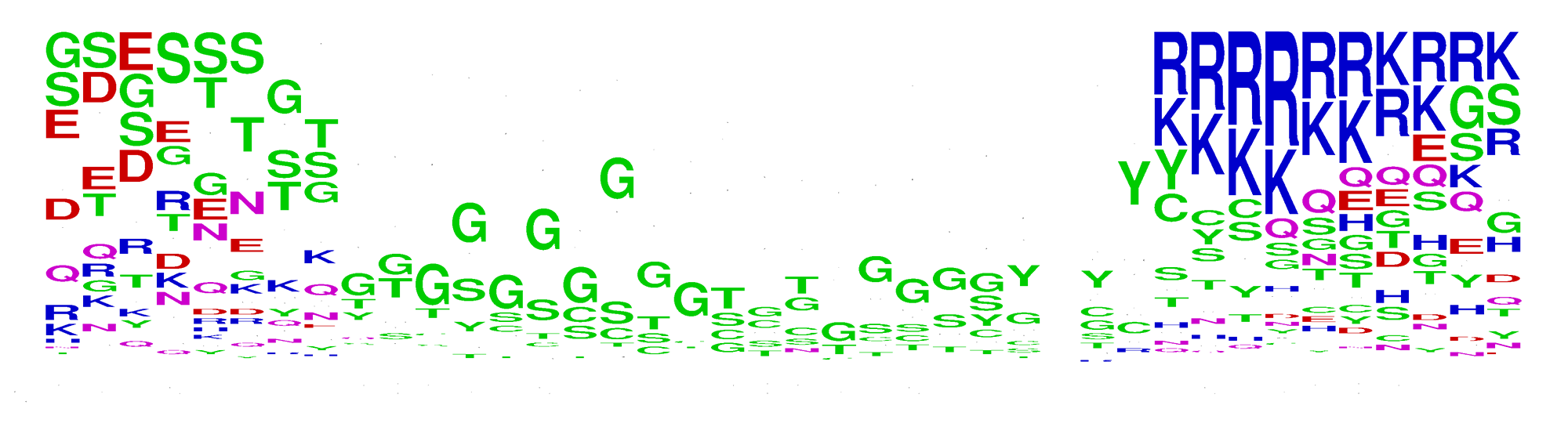
The features of GS-SMD server:
- steered molecular dynamics of all known substrates of GS in the substrate-GS complex employing the implicit environments approach.
- possibility to introduce mutations in the substrate and in GS.
- full control of SMD parameters (direction of pulling, speed, and spring constant).
- possibility to select three systems: full atomistic model GS complex, its membranous part, or the presenilin, the catalytic subunit of GS.
- wide set of analyses for the single trajectories and for their comparisons.
This website is free and open to all users and there is no login requirement
This website do not use any tracking cookies.
